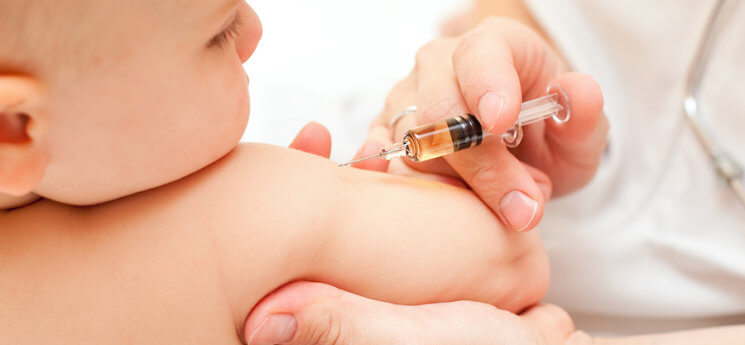
The American Academy of Pediatrics (AAP) recommends all children ages 6 months or older be immunized against influenza as soon as vaccine is available, either with the trivalent vaccine that protects against three strains of the virus, or with a new quadrivalent vaccine that protects against four strains.
Children Should be Vaccinated Against Influenza
The AAP released its updated recommendations in a policy statement, Recommendations for Prevention and Control of Influenza in Children, 2019-2020.
A special effort should be made to vaccinate people in vulnerable groups, including children with chronic health conditions, children of American Indian or Alaskan Native heritage, health care workers, women who are pregnant, may become pregnant or are breastfeeding, and household contacts and caregivers of children in high-risk populations.
“Parents should not delay vaccinating their children to obtain a specific vaccine,” said pediatrician Henry Bernstein, D.O., FAAP, the lead author of the flu recommendations. “Influenza virus is unpredictable, and what’s most important is that people receive the vaccine soon, so that they will be protected when the virus begins circulating.”
Recent data show that most people who have an egg allergy can receive the inactivated influenza vaccine. Although inactivated vaccine given as a single, age-appropriate dose is well-tolerated by all recipients who have egg allergy, pediatricians should consult with an allergist for any child with history of a severe reaction.
The AAP continues to recommend using the antiviral medicines oseltamivir or zanamivir for children hospitalized for influenza or with severe influenza symptoms, and in children with influenza who have underlying chronic conditions.
Given its known safety profile, oseltamivir can be used to treat influenza in both term and preterm infants from birth, while chemoprophylaxis should only be considered in term infants.